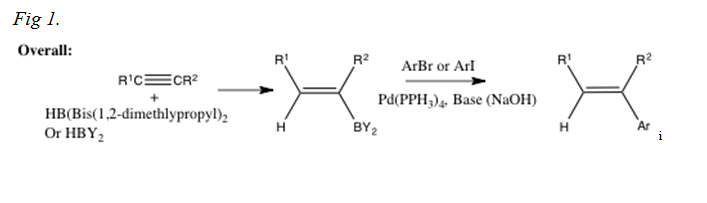
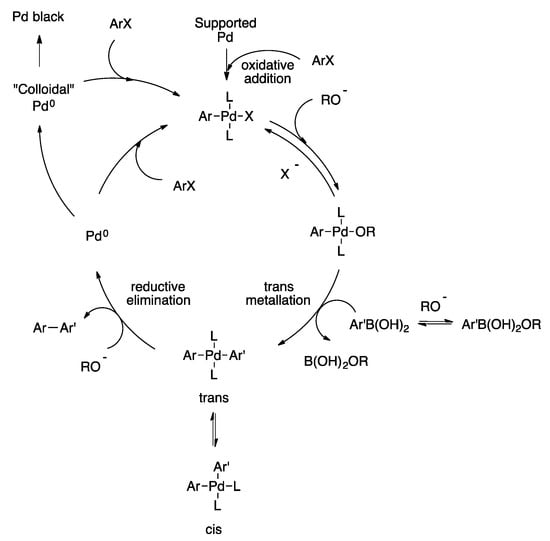
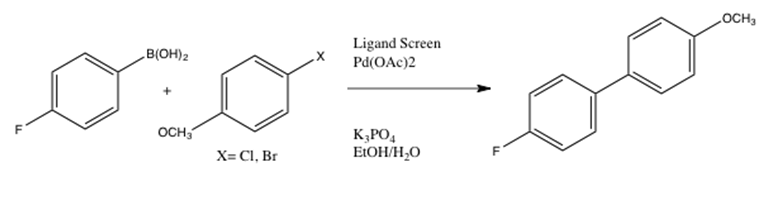
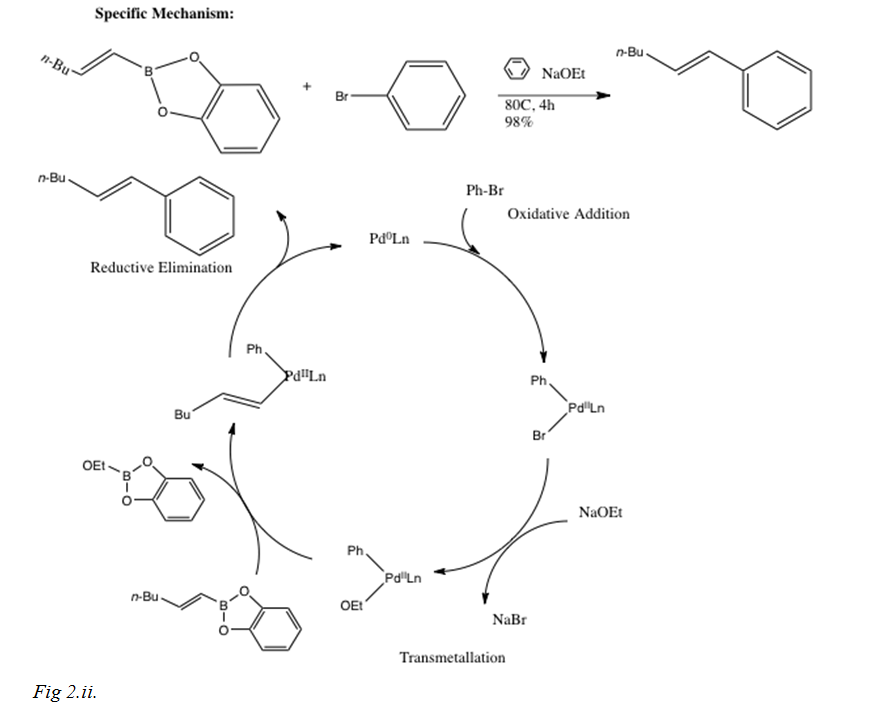
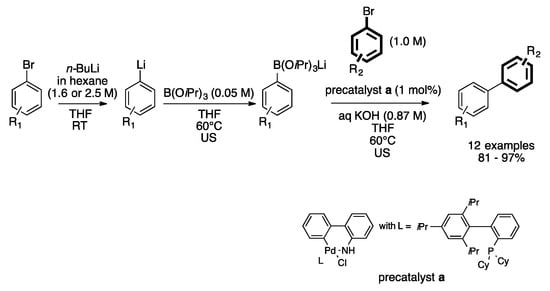
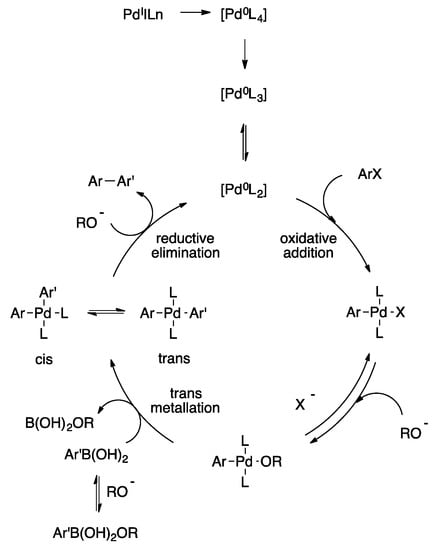
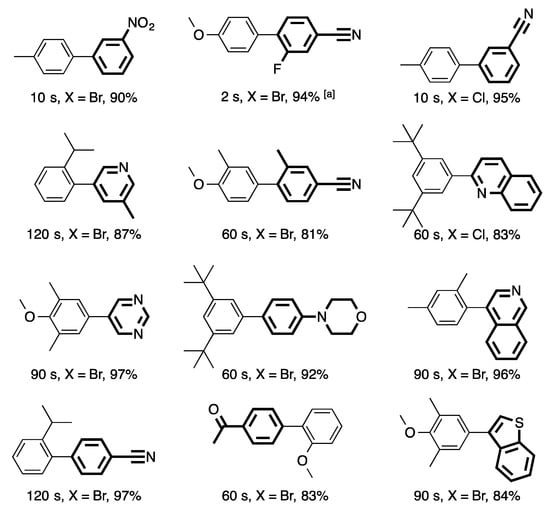
Palladium-Catalyzed Suzuki−Miyaura Cross-Coupling Reactions Employing Dialkylbiaryl Phosphine Ligands | Accounts of Chemical Research
Recent advances in the Suzuki–Miyaura cross-coupling reaction using efficient catalysts in eco-friendly media - Green Chemistry (RSC Publishing) DOI:10.1039/C8GC02860E

Palladium-Catalyzed Suzuki−Miyaura Cross-Coupling Reactions Employing Dialkylbiaryl Phosphine Ligands | Accounts of Chemical Research

Palladium-Catalyzed Suzuki−Miyaura Cross-Couplings of Sulfonyl Chlorides and Boronic Acids | Organic Letters

Palladium-Catalyzed Suzuki−Miyaura Cross-Coupling Reactions Employing Dialkylbiaryl Phosphine Ligands | Accounts of Chemical Research

Palladium-mediated Suzuki-Miyaura Cross-Coupling Reaction of Potassium Boc-protected aminomethyltrifluoroborate with DNA-Conjugated aryl bromides for DNA-Encoded chemical library synthesis - ScienceDirect

Palladium-Catalyzed Suzuki−Miyaura Cross-Coupling Reactions Employing Dialkylbiaryl Phosphine Ligands | Accounts of Chemical Research
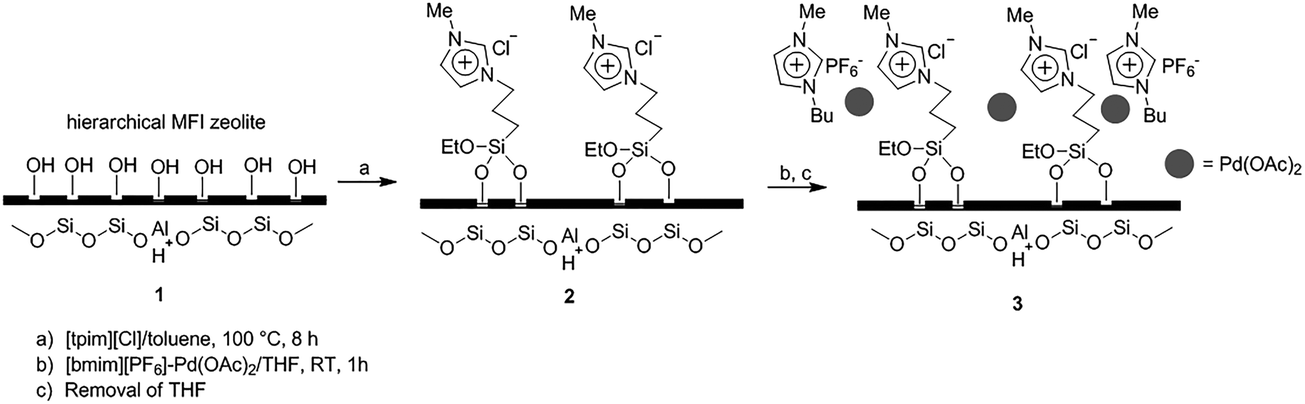
Recent advances in the Suzuki–Miyaura cross-coupling reaction using efficient catalysts in eco-friendly media - Green Chemistry (RSC Publishing) DOI:10.1039/C8GC02860E
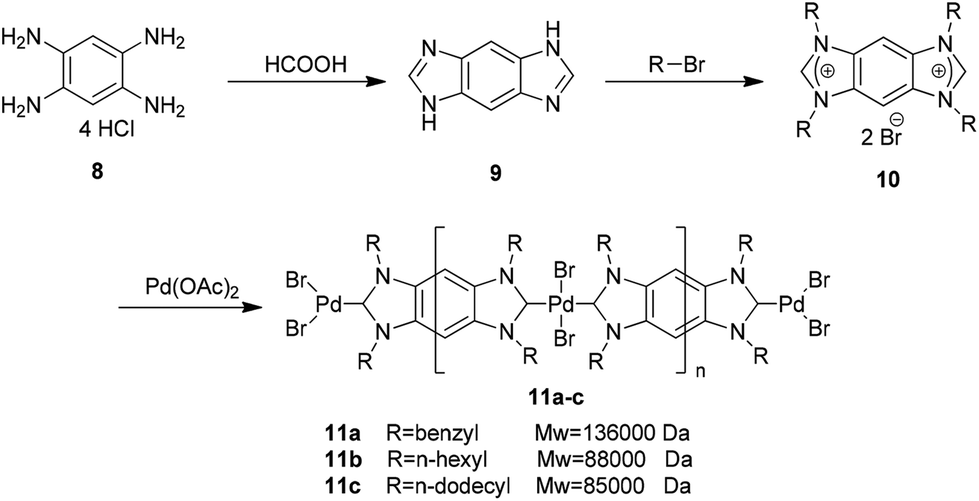
![Catalysts | Free Full-Text | Palladium PEPPSI-IPr Complex Supported on a Calix[8]arene: A New Catalyst for Efficient Suzuki–Miyaura Coupling of Aryl Chlorides Catalysts | Free Full-Text | Palladium PEPPSI-IPr Complex Supported on a Calix[8]arene: A New Catalyst for Efficient Suzuki–Miyaura Coupling of Aryl Chlorides](https://www.mdpi.com/catalysts/catalysts-10-01081/article_deploy/html/images/catalysts-10-01081-g001-550.jpg)
Catalysts | Free Full-Text | Palladium PEPPSI-IPr Complex Supported on a Calix[8]arene: A New Catalyst for Efficient Suzuki–Miyaura Coupling of Aryl Chlorides

Simple Palladium(II) Precatalyst for Suzuki−Miyaura Couplings: Efficient Reactions of Benzylic, Aryl, Heteroaryl, and Vinyl Coupling Partners | Organic Letters

Palladium-Catalyzed Suzuki−Miyaura Cross-Coupling Reactions Employing Dialkylbiaryl Phosphine Ligands | Accounts of Chemical Research